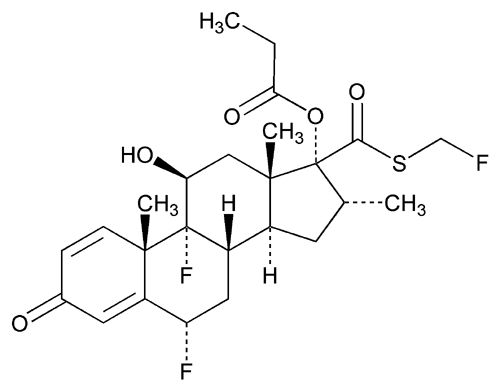
Fluticasone Propionate
» Fluticasone Propionate contains not less than 98.0 percent and not more than 101.0 percent of C25H31F3O5S, calculated on the anhydrous, solvent-free basis.
Packaging and storage—
Preserve in tight, light-resistant containers, and store at a temperature not exceeding 30 .
.
Labeling—
Fluticasone Propionate in the form of microcrystals is so labeled.
USP Reference standards  11
11 —
—
USP Fluticasone Propionate RS.
USP Fluticasone Propionate System Suitability Mixture RS .
.
USP Fluticasone Propionate Resolution Mixture RS.
USP Fluticasone Propionate RS.
USP Fluticasone Propionate System Suitability Mixture RS
 .
.
USP Fluticasone Propionate Resolution Mixture RS.
Identification—
B:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
Specific rotation  781S
781S :
between +32
:
between +32 and +36
and +36 (t = 20
(t = 20 ), calculated on the anhydrous, solvent-free basis.
), calculated on the anhydrous, solvent-free basis.
Test solution:
0.5% (w/v) of Fluticasone Propionate in dichloromethane (0.5 g in 100 mL).
Water, Method I  921
921 :
not more than 0.2% (w/w).
:
not more than 0.2% (w/w).
Related compounds—
Solution A—
Mix 0.5 mL of phosphoric acid in 1000 mL of acetonitrile.
Solution B—
Mix 0.5 mL of phosphoric acid in 1000 mL of methanol.
Solution C—
Mix 0.5 mL of phosphoric acid in 1000 mL of water.
Mobile phase—
Use variable mixtures of Solution A, Solution B, and Solution C, as directed under Chromatographic system. Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
System suitability solution—
Transfer approximately 2.0 mg of USP Fluticasone Propionate System Suitability Mixture RS to 5 mL of Solution A, and sonicate to dissolve. Add 5 mL of Solution C, and mix.
Test solution—
Transfer approximately 2.0 mg of Fluticasone Propionate to 5 mL of Solution A, and sonicate to dissolve. Add 5 mL of Solution C, and mix.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 239-nm detector and a 4.6-mm × 25-cm column that contains 5-µm packing L1. The flow rate is about 1.0 mL per minute. The column temperature is maintained at 40
)—
The liquid chromatograph is equipped with a 239-nm detector and a 4.6-mm × 25-cm column that contains 5-µm packing L1. The flow rate is about 1.0 mL per minute. The column temperature is maintained at 40 . The chromatograph is programmed (see Table 1), as follows.
. The chromatograph is programmed (see Table 1), as follows.
Chromatograph the System suitability solution, and measure the peak responses as directed for Procedure: the resolution, R, between fluticasone propionate related compound B and fluticasone propionate related compound C is not less than 0.6; the resolution, R, between fluticasone propionate related compound D and fluticasone propionate is not less than 1.5; the relative retention times and limits are as provided in Table 2.
Table 1
| Time (minutes) |
Solution
A (v) |
Solution
B (v) |
Solution
C (v) |
Elution |
| 0 | 42 | 3 | 55 | equilibrium |
| 0–40 | 42®53 | 3 | 55®44 | linear gradient |
| 40–60 | 53®87 | 3 | 44®10 | linear gradient |
| 60–70 | 87 | 3 | 10 | isocratic |
| 70–75 | 87®42 | 3 | 10®55 | re-equilibration |
Procedure—
Inject a volume (about 50 µL) of the Test solution into the chromatograph, record the chromatograms, and measure all of the peak responses. Calculate the percentage of each impurity in the portion of Fluticasone Propionate taken by the formula:
100(ri / rs)
in which ri is the peak response for each impurity; and rs is the sum of the responses of all the peaks.
Table 2
| Compound | Approximate Relative Retention Time | Limit (%) |
| Fluticasone propionate related compound A | 0.5 | 0.2 |
| Fluticasone propionate related compound B | 0.75 | 0.1 |
| Fluticasone propionate related compound C | 0.8 | 0.1 |
| Fluticasone propionate related compound D | 0.95 | 0.3 |
| Fluticasone propionate | 1.0 | — |
| Fluticasone propionate related compound E | 1.3 | 0.3 |
| Other impurities | — | 0.1 |
| Total impurities* | — | 1.0 |
|
*
Calculate the total impurities from the sum of all impurity peaks greater than or equal to 0.05%.
|
||
Limit of acetone—
Internal standard solution—
Prepare a 0.05% (v/v) solution of tetrahydrofuran in dimethylformamide.
Standard solution—
Prepare 0.05% (v/v) of acetone in Internal standard solution.
Test solution—
Dissolve an accurately weighed quantity of Fluticasone Propionate in Internal standard solution to obtain a solution having a known concentration of about 50 mg per mL.
Chromatographic system (see Chromatography  621
621 )—
The gas chromatograph is equipped with a flame-ionization detector and a 0.53-mm × 25-m column coated with a 2-µm film of phase G15. The carrier gas is nitrogen or helium, flowing at a rate of about 5.5 mL per minute. The column temperature is programmed as follows. Initially the temperature of the column is equilibrated at 60
)—
The gas chromatograph is equipped with a flame-ionization detector and a 0.53-mm × 25-m column coated with a 2-µm film of phase G15. The carrier gas is nitrogen or helium, flowing at a rate of about 5.5 mL per minute. The column temperature is programmed as follows. Initially the temperature of the column is equilibrated at 60 for 3.5 minutes, then the temperature is increased at the rate of 30
for 3.5 minutes, then the temperature is increased at the rate of 30 per minute to 180
per minute to 180 , and maintained at 180
, and maintained at 180 for 3 minutes. The splitless injector temperature is maintained at 150
for 3 minutes. The splitless injector temperature is maintained at 150 , and the detector temperature is maintained at 250
, and the detector temperature is maintained at 250 . Chromatograph the Standard solution, and record the peak responses as directed for Procedure: the relative standard deviation for replicate injections of the Standard solution is not more than 5.0%.
. Chromatograph the Standard solution, and record the peak responses as directed for Procedure: the relative standard deviation for replicate injections of the Standard solution is not more than 5.0%.
Procedure—
Separately inject equal volumes (about 0.1 µL) of the Standard solution and the Test solution into the chromatograph, and record the peak responses. Calculate the percentage of acetone (% w/w) in the portion of Fluticasone Propionate taken by the formula:
 ; C is the concentration, in g per mL, of Fluticasone Propionate in the Test solution; and RU and RS are the ratios of the acetone peak response to the tetrahydrofuran peak response obtained from the Test solution and the Standard solution, respectively: not more than 1.0% (w/w) is found.
; C is the concentration, in g per mL, of Fluticasone Propionate in the Test solution; and RU and RS are the ratios of the acetone peak response to the tetrahydrofuran peak response obtained from the Test solution and the Standard solution, respectively: not more than 1.0% (w/w) is found.
0.05D/C(RU / RS)
in which D is the density of acetone at 20
Assay—
0.01 M Monobasic ammonium phosphate buffer, pH 3.5—
Dissolve 1.15 g of monobasic ammonium phosphate in 1000 mL of water, adjust with phosphoric acid to a pH of 3.5 ± 0.05, and mix.
Mobile phase—
Prepare a filtered and degassed mixture of methanol, 0.01 M Monobasic ammonium phosphate buffer, pH 3.5, and acetonitrile (50:35:15). Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Resolution solution—
Dissolve approximately 2.5 mg of USP Fluticasone Propionate Resolution Mixture RS in 50 mL of Mobile phase.
Standard preparation—
Dissolve an accurately weighed quantity of USP Fluticasone Propionate RS in Mobile phase, and dilute quantitatively, and stepwise if necessary, to obtain a solution having a known concentration of about 0.04 mg per mL.
Assay preparation—
Dissolve an accurately weighed quantity of Fluticasone Propionate in Mobile phase, and dilute quantitatively, and stepwise if necessary, to obtain a solution having a known concentration of about 0.04 mg per mL.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 239-nm detector and a 4.6-mm × 25-cm column that contains 5-µm packing L1. The flow rate is about 1.5 mL per minute. The column temperature is maintained at 40
)—
The liquid chromatograph is equipped with a 239-nm detector and a 4.6-mm × 25-cm column that contains 5-µm packing L1. The flow rate is about 1.5 mL per minute. The column temperature is maintained at 40 . Chromatograph the Resolution solution, and record the peak areas as directed for Procedure: the relative retention times are about 1.10 for fluticasone propionate related compound D and 1.0 for fluticasone propionate; and the resolution, R, between fluticasone propionate and fluticasone propionate related compound D is not less than 1.5. Chromatograph the Standard solution, and record the peak areas as directed for Procedure: the relative standard deviation for replicate injections of the Standard solution is not more than 2%.
. Chromatograph the Resolution solution, and record the peak areas as directed for Procedure: the relative retention times are about 1.10 for fluticasone propionate related compound D and 1.0 for fluticasone propionate; and the resolution, R, between fluticasone propionate and fluticasone propionate related compound D is not less than 1.5. Chromatograph the Standard solution, and record the peak areas as directed for Procedure: the relative standard deviation for replicate injections of the Standard solution is not more than 2%.
Procedure—
Separately inject equal volumes (about 20 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the areas for the major peaks. Calculate the quantity, in mg, of C25H31F3O5S in the portion of Fluticasone Propionate taken by the formula:
CV(rU / rS)
in which C is the concentration of USP Fluticasone Propionate RS, in mg per mL, in the Standard preparation; V is the volume, in mL, of the Assay preparation; and rU and rS are the peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Kahkashan Zaidi, Ph.D.
Senior Scientist 1-301-816-8269 |
(AER05) Aerosols05 |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2434
Pharmacopeial Forum: Volume No. 32(2) Page 337
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.