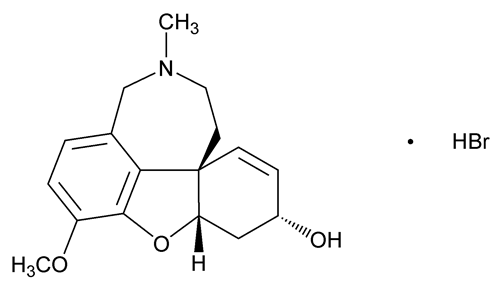
Galantamine Hydrobromide

 231
231 :
:
 (Official 1-Dec-2015)
(Official 1-Dec-2015)
(ga lan' ta meen hye'' droe broe' mide).
DEFINITION
Galantamine Hydrobromide contains NLT 98.0% and NMT 102.0% of galantamine hydrobromide (C17H21NO3·HBr), calculated on the dried basis.
IDENTIFICATION
• A. Infrared Absorption  197K
197K
[Note—Specimens are to be prepared using undried USP Galantamine Hydrobromide RS and the test article. ]
• B.
The retention time of the major peak of the Sample solution corresponds to that of the System suitability solution, as obtained in the Assay.
• C. Identification Tests—General, Bromide  191
191
Sample solution:
A solution of 7 mg/mL in water
Acceptance criteria:
Meets the requirements of the silver nitrate precipitate test
ASSAY
• Procedure
Diluent:
Methanol and water (1:19)
Buffer:
0.79 g/L of dibasic sodium phosphate dihydrate and 2.46 g/L of anhydrous monobasic sodium phosphate in water
Solution A:
Methanol and Buffer (5:95)
Solution B:
Acetonitrile
Mobile phase:
See Table 1.
Table 1
| Time (min) |
Solution A (%) |
Solution B (%) |
|---|---|---|
| 0 | 100 | 0 |
| 6.0 | 100 | 0 |
| 20.0 | 95 | 5 |
| 35.0 | 85 | 15 |
| 50.0 | 80 | 20 |
| 51.0 | 40 | 60 |
| 55.0 | 40 | 60 |
| 56.0 | 100 | 0 |
| 60.0 | 100 | 0 |
System suitability solution:
1 mg/mL of USP Galantamine Hydrobromide Related Compounds Mixture RS in Diluent
Standard solution:
1.0 mg/mL of USP Galantamine Hydrobromide RS in Diluent
Sample solution:
1.0 mg/mL of Galantamine Hydrobromide in Diluent
Chromatographic system
Mode:
LC
Detector:
UV 230 nm
Column:
4.6-mm × 10-cm; 3.5-µm packing L1
Column temperature:
55
Flow rate:
1.5 mL/min
Injection volume:
20 µL
System suitability
Samples:
System suitability solution and Standard solution
[Note—For relative retention times, see Table 2. ]
Suitability requirements
Resolution:
NLT 4.5 between galantamine and 6 -hexahydrogalantamine, System suitability solution
-hexahydrogalantamine, System suitability solution
Tailing factor:
NMT 2.0 for galantamine, System suitability solution
Relative standard deviation:
NMT 1.0%, Standard solution
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of galantamine hydrobromide (C17H21NO3·HBr) in the portion of Galantamine Hydrobromide taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Galantamine Hydrobromide RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Galantamine Hydrobromide in the Sample solution (mg/mL) |
Acceptance criteria:
98.0%–102.0% on the dried basis
IMPURITIES
• Residue on Ignition  281
281 :
NMT 0.1%
:
NMT 0.1%
Delete the following:
• Limit of Palladium
[Note—Perform this test only if palladium is a known inorganic impurity of the manufacturing process. ]
Standard stock solution:
20 mg/L of palladium reference stock solution (NIST traceable) in water
Aqua regia:
Under a hood, carefully mix hydrochloric acid and nitric acid (3:1).
[Note—To obtain each of the required Standard solutions, it is recommended that the required volume of Standard stock solution be mixed with a volume of Aqua regia equivalent to 5% of the final volume, followed by water. ]
Standard solution A:
0.2 mg/L of palladium from the Standard stock solution in water
Standard solution B:
1.0 mg/L of palladium from the Standard stock solution in water
Standard solution C:
2.0 mg/L of palladium from the Standard stock solution in water
System suitability solution:
Prepare a solution having a known concentration of 1.6 mg/L of palladium, as directed for Standard solutions.
Sample solution:
Weigh 1 g of Galantamine Hydrobromide. Transfer the sample to an appropriate digestion system, and digest using appropriate acids (e.g., nitric acid or mixtures of nitric acid and sulfuric acid and mixtures of nitric acid and hydrogen peroxide). After digestion, heat to dryness. Add 0.5 mL of Aqua regia and 2 mL of water. Warm gently to dissolve any residue. Allow to cool. Transfer quantitatively to a 10-mL volumetric flask, and dilute with water to volume.
Digestion blank solution:
Prepare this solution following the procedure for the Sample solution, without the test article.
Instrumental conditions
Mode:
Atomic absorption spectroscopy (flame)
Analytical wavelength:
247.6 nm (0.2-nm slit width)
Lamp:
Palladium hollow-cathode
Blank solution:
Dilute 5 mL of Aqua regia with water to 100 mL.
System suitability
Samples:
Standard solution A, Standard solution B, Standard solution C, System suitability solution, and Blank solution
Using the Standard solutions and Blank solution, construct a calibration curve.
Suitability requirements
Correlation coefficient:
NLT 0.99
Recovery:
87.5%–112.5%, System suitability solution. [Note—Recovery is calculated using the calibration curve. ]
Analysis
Samples:
Sample solution and Digestion blank solution
Calculate the concentration of palladium in the Sample solution, using the calibration curve, corrected for the Digestion blank solution and the sample weight. Calculate the amount of palladium in the Galantamine Hydrobromide taken to prepare the Sample solution.
Acceptance criteria:
NMT 10 ppm
• Organic Impurities
Diluent, Buffer, Solution A, Solution B, Mobile phase, System suitability solution, Sample solution, Chromatographic system, and System suitability:
Prepare as directed in the Assay.
Standard solution:
5.0 µg/mL of USP Galantamine Hydrobromide RS in Diluent
Analysis
Samples:
Sample solution and Standard solution
[Note—Ignore the peak due to bromide near the void volume and any peak below 0.05%. ]
Calculate the percentage of each impurity in the portion of Galantamine Hydrobromide taken, on the dried basis:
Result = (rU/rS) × (CS/CU) × (1/F) × (100/100  L)
L)
| rU | = | = peak response of each impurity from the Sample solution |
| rS | = | = peak response of galantamine from the Standard solution |
| CS | = | = concentration of USP Galantamine Hydrobromide RS in the Standard solution (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) |
| F | = | = relative response factor (see Table 2) |
| L | = | = loss on drying in percent |
Acceptance criteria:
See Table 2.
Table 2
| Name | Relative Retention Time |
Relative Response Factor |
Acceptance Criteria, NMT (%) |
|---|---|---|---|
| N-Desmethyl galantaminea | 0.29 | 1.2 | 0.6 |
| O-Desmethyl galantamineb | 0.35 | 1.1 | 0.20 |
| Galantamine N- oxidec |
0.65 | 0.96 | 0.20 |
| Dihydrogalantamined | 0.82 | 0.81 | 0.35 |
| Galantamine | 1.00 | 1.0 | — |
| 6S-Galantaminee | 1.16 | 0.95 | 0.20 |
| Narwedinef | 1.64 | 1.9 | 0.15 |
| Didehydrodeoxygalantamineg | 2.05 | 1.2 | 0.40 |
| Any unspecified impurity |
— | 1.0 | 0.10 |
| Total impuritiesh | — | — | 1.0 |
|
a
(4aS,6R,8aS)-4a,5,9,10,11,12-Hexahydro-3-methoxy-6H-benzofuro[3a,3,2-ef][2]benzazepin-6-ol.
b
(4aS,6R,8aS)-4a,5,9,10,11,12-Hexahydro-3-hydroxy-11-methyl-6H-benzofuro[3a,3,2-ef][2]benzazepin-6-ol.
c
(4aS,6R,8aS)-4a,5,9,10,11,12-Hexahydro-3-methoxy-11-methyl-6H-benzofuro[3a,3,2-ef][2]benzazepin-6-ol, N-oxide; also known as 6
d
(4aS,6R,8aS)-4a,5,7,8,9,10,11,12-Octahydro-3-methoxy-11-methyl-6H-benzofuro[3a,3,2-ef][2]benzazepin-6-ol.; also known as 6
e
(4aS,6S,8aS)-4a,5,9,10,11,12-Hexahydro-3-methoxy-11-methyl-6H-benzofuro[3a,3,2-ef][2]benzazepin-6-ol; also known as 6
f
(4aS,8aS)-4a,5,9,10,11,12-Hexahydro-3-methoxy-11-methyl-6H-benzofuro[3a,3,2-ef][2]benzazepin-6-one. [Note—This is a process impurity that may be found in galantamine hydrobromide isolated from a natural source. ]
g
(4aS,8aS)-9,10,11,12-Tetrahydro-3-methoxy-11-methyl-6H-benzofuro[3a,3,2-ef][2]benzazepine; also known as tetrahydrogalantamine.
h
Do not include the 4R,8R-stereoisomer.
|
|||
• Enantiomeric Purity
[Note—If Galantamine Hydrobromide is not isolated from a natural source, perform either Procedure 1 or Procedure 2. ]
Procedure 1
Background electrolyte solution:
8.9 g/L of dibasic sodium phosphate dihydrate in water. Adjust with phosphoric acid to a pH of 3.0.
Run buffer:
19.6 g/L of  -cyclodextrin hydrate in Background electrolyte solution. Pass the solution through a filter of 0.22-µm pore size.
-cyclodextrin hydrate in Background electrolyte solution. Pass the solution through a filter of 0.22-µm pore size.
Standard solution:
5 µg/mL of USP Galantamine Hydrobromide Racemic RS in water. Pass the solution through a filter of 0.22-µm pore size, discarding the first 8 mL.
Sample solution:
0.5 mg/mL of Galantamine Hydrobromide in water. Pass the solution through a filter of 0.22-µm pore size, discarding the first 8 mL.
Capillary rinse procedure:
Use separate Run buffer vials for the capillary rinse and sample analysis. Proceed as directed in Table 3.
Table 3
| Step # | Solution/Gas | Time (min) |
|---|---|---|
| 1 | 0.1 N sodium hydroxide | 15 |
| 2 | Water | 10 |
| 3 | Suitable gas | 5 |
[Note—If a new or dry capillary is being used, rinse with 1 N sodium hydroxide for 30 min, followed by rinsing with water for 15 min. Dry it with air or nitrogen for 10 min. ]
Electrophoretic system
Mode:
CE
Detector:
UV 214 nm
Column:
75-µm × 60-cm uncoated fused-silica
Column temperature:
20
Applied voltage:
250 V/cm, positive polarity
Run time:
35 min
System suitability
Sample:
Standard solution. [Note—For the purpose of identification, the 4S,8S stereoisomer elutes at an approximate relative migration time (RMT) of 1.00, and the 4R,8R stereoisomer elutes at an RMT of about 1.05. ]
Suitability requirements
Resolution:
NLT 2.5 between the two enantiomers
Relative standard deviation:
NMT 10% for the 4R,8R stereoisomer peak
Measure the migration times and peak responses: the migration times for the 4R,8R stereoisomer in the electropherograms for the Sample solution should not deviate by more than 5% of the migration time for the same component in the electropherogram of the Standard solution.
Analysis
Samples:
Standard solution and Sample solution
Injection:
[Note—Rinse the capillary between injections as follows: water for 5 min, followed by Run buffer for 5 min. Rinse times are based on a rinse pressure of 1.4 bar. ]
Sample solution:
34.5 mbar for 4 s
Run buffer:
6.9 mbar for 5 s
Calculate the corrected peak responses using the formula:
Result = (r/m)
| r | = | = peak response |
| m | = | = migration time of the peak (min) |
Calculate the limit of the 4R,8R isomer, in percent, in the portion of Galantamine Hydrobromide taken:
Result = (rCU/rCS) × (CS/CU) × P × 100
| rCU | = | = average corrected peak responses for the 4R,8R isomer from the Sample solution |
| rCS | = | = average corrected peak responses for the 4R,8R isomer from the Standard solution |
| CS | = | = concentration of USP Galantamine Hydrobromide Racemic RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Galantamine Hydrobromide in the Sample solution (mg/mL) |
| P | = | = chiral purity of USP Galantamine Hydrobromide Racemic RS, 0.5 |
Acceptance criteria:
NMT 0.10% of the 4R,8R stereoisomer
Procedure 2
[Note—Use low-actinic glassware and vials. It is recommended that precautions be taken to protect all solutions from light. ]
Buffer:
8.2 g/L of sodium acetate in water
Mobile phase:
Acetonitrile and Buffer (2:98). Adjust with acetic acid to a pH of 6.5.
System suitability solution:
2.4 µg/mL of USP Galantamine Hydrobromide Racemic RS in water. [Note—This solution will contain about 1.2 µg/mL of the 4R,8R stereoisomer. ]
Sample solution:
1.2 mg/mL of Galantamine Hydrobromide in water
Chromatographic system
Mode:
LC
Detector:
UV 230 nm
Column:
4.0-mm × 15-cm; 5-µm packing L41. [Note—Alternatively a 2.0-mm × 15.0-cm column containing 5-µm L41 packing can be used with a recommended flow rate of about 0.2 mL/min. ]
Flow rate:
0.8 mL/min
Injection volume:
5 µL
System suitability
Sample:
System suitability solution. [Note—The 4R,8R stereoisomer elutes first as the minor peak followed by the major peak due to galantamine (which is the same as the 4S,8S stereoisomer). ]
Suitability requirements
Resolution:
NLT 3.0 between the 4R,8R stereoisomer and galantamine peaks
Relative standard deviation:
NMT 5.0% for the 4R,8R stereoisomer peak
Analysis
Sample:
Sample solution
Calculate the percentage of 4R,8R stereoisomer in the portion of Galantamine Hydrobromide taken:
Result = 100 × [r4R,8R/(r4R,8R + r4S,8S)]
| r4R,8R | = | = peak area of the 4R,8R stereoisomer from the Sample solution |
| r4S,8S | = | = peak area of the galantamine peak from the Sample solution |
Acceptance criteria:
NMT 0.10% of the 4R,8R stereoisomer
SPECIFIC TESTS
• Optical Rotation, Specific Rotation  781
781
[Note—If Galantamine Hydrobromide is isolated from a natural source, perform the test for Optical Rotation. ]
Sample solution:
20 mg/mL in water
Acceptance criteria:
 90
90 to
to  100
100
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Store at room temperature. Preserve in well-closed containers.
• Labeling:
Label it to state if the source is naturally derived or is synthetic. If the source is not natural, perform either Procedure 1 or Procedure 2 of the test for Enantiomeric Purity. If the source is natural, perform the test for Optical Rotation  781
781 , Specific Rotation.
, Specific Rotation.
• USP Reference Standards  11
11
USP Galantamine Hydrobromide Related Compounds Mixture RS
Contains galantamine hydrobromide, 6 -hexahydrogalantamine, 6
-hexahydrogalantamine, 6 -octahydrogalantamine, 6
-octahydrogalantamine, 6 -hexahydrogalantamine, and tetrahydrogalantamine.
-hexahydrogalantamine, and tetrahydrogalantamine.
Contains galantamine hydrobromide, 6
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Heather R. Joyce, Ph.D.
Associate Scientific Liaison (301) 998-6792 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP38–NF33 Page 3646
Pharmacopeial Forum: Volume No. 38(1)